CHEMICAL KINETICS
Table of Contents
From baking a cake to determining the useful lifespan of a bridge, rates of chemical reactions play important roles in our understanding of processes that involve chemical changes. Two questions are typically posed when planning to carry out a chemical reaction. The first is: “Will the reaction produce the desired products in useful quantities?” The second question is: “How rapidly will the reaction occur?” A third question is often asked when investigating reactions in greater detail: “What specific molecular-level processes take place as the reaction occurs?” Knowing the answer to this question is of practical importance when the yield or rate of a reaction needs to be controlled.
The study of chemical kinetics concerns the second and third questions—that is, the rate at which a reaction yields products and the molecular-scale means by which a reaction occurs
Chemical Reaction Rates
A rate is a measure of how some property varies with time. Speed is a familiar rate that expresses the distance traveled by an object in a given amount of time. Wage is a rate that represents the amount of money earned by a person working for a given amount of time. Likewise, the rate of a chemical reaction is a measure of how much reactant is consumed, or how much product is produced, by the reaction in a given amount of time.
The rate of reaction is the change in the amount of a reactant or product per unit time. Reaction rates are therefore determined by measuring the time dependence of some property that can be related to reactant or product amounts. Rates of reactions that consume or produce gaseous substances, for example, are conveniently determined by measuring changes in volume or pressure. For reactions involving one or more colored substances, rates may be monitored via measurements of light absorption. For reactions involving aqueous electrolytes, rates may be measured via changes in a solution’s conductivity
For reactants and products in solution, their relative amounts (concentrations) are conveniently used for purposes of expressing reaction rates. For example, the concentration of hydrogen peroxide, H2O2, in an aqueous solution changes slowly over time as it decomposes according to the equation
2H2O2 (aq) ———> 2H2O (l) + O2 (g)
The rate at which the hydrogen peroxide decomposes can be expressed in terms of the rate of change of its concentration, as shown here

This mathematical representation of the change in species concentration over time is the rate expression for the reaction. The brackets indicate molar concentrations, and the symbol delta (Δ) indicates “change in.” Thus [H2 O2]t1 represents the molar concentration of hydrogen peroxide at some time t1; likewise [H2 O2]t2 represents the molar concentration of hydrogen peroxide at some time t2 and Δ[H2O2] represents the change in molar concentration of hydrogen peroxide during the time interval Δt (that is, t2 − t1). Since the reactant concentration decreases as the reaction proceeds, Δ[H2O2] is a negative quantity. Reaction rates are, by convention, positive quantities, and so this negative change in concentration is multiplied by −1. The table below provides an example of data collected during the decomposition of H2O2.

The rate of decomposition of H2O2 in an aqueous solution decreases as the concentration of H2O2 decreases.
To obtain the tabulated results for this decomposition, the concentration of hydrogen peroxide was measured every 6 hours over the course of a day at a constant temperature of 40 °C. Reaction rates were computed for each time interval by dividing the change in concentration by the corresponding time increment, as shown here for the first 6-hour period

This behavior indicates the reaction continually slows with time. Using the concentrations at the beginning and end of a time period over which the reaction rate is changing results in the calculation of an average rate for the reaction over this time interval. At any specific time, the rate at which a reaction is proceeding is known as its instantaneous rate. The instantaneous rate of a reaction at “time zero,” when the reaction commences, is its initial rate. Consider the analogy of a car slowing down as it approaches a stop sign. The vehicle’s initial rate—analogous to the beginning of a chemical reaction—would be the speedometer reading at the moment the driver begins pressing the brakes (t0). A few moments later, the instantaneous rate at a specific moment—call it t1—would be somewhat slower, as indicated by the speedometer reading at that point in time. As time passes, the instantaneous rate will continue to fall until it reaches zero, when the car (or reaction) stops. Unlike instantaneous speed, the car’s average speed is not indicated by the speedometer; but it can be calculated as the ratio of the distance traveled to the time required to bring the vehicle to a complete stop (Δt). Like the decelerating car, the average rate of a chemical reaction will fall somewhere between its initial and final rates.
The instantaneous rate of a reaction may be determined one of two ways. If experimental conditions permit the measurement of concentration changes over very short time intervals, then average rates computed as described earlier provide reasonably good approximations of instantaneous rates. Alternatively, a graphical procedure may be used that, in effect, yields the results that would be obtained if short time interval measurements were possible. In a plot of the concentration of hydrogen peroxide against time, the instantaneous rate of decomposition of H2O2 at any time t is given by the slope of a straight line that is tangent to the curve at that time (Below Graph). These tangent line slopes may be evaluated using calculus
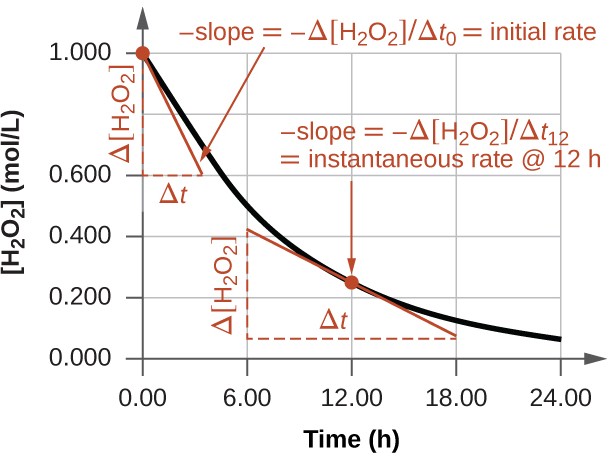
This graph shows a plot of concentration versus time for a 1.000 M solution of H2O2. The rate at any time is equal to the negative of the slope of a line tangent to the curve at that time. Tangents are shown at t = 0 h (“initial rate”) and at t = 12 h (“instantaneous rate” at 12 h)
Relative Rates of Reaction
The rate of a reaction may be expressed as the change in concentration of any reactant or product. For any given reaction, these rate expressions are all related simply to one another according to the reaction stoichiometry. The rate of the general reaction
aA —-à bB
can be expressed in terms of the decrease in the concentration of A or the increase in the concentration of B. These two rate expressions are related by the stoichiometry of the reaction:

Consider the reaction represented by the following equation:
2NH3 (g) —–à N2 (g) + 3 H2 (g)
The relation between the reaction rates expressed in terms of nitrogen production and ammonia consumption, for example, is:
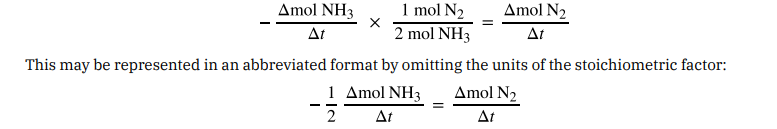
Note that a negative sign has been included as a factor to account for the opposite signs of the two amount changes (the reactant amount is decreasing while the product amount is increasing). For homogeneous reactions, both the reactants and products are present in the same solution and thus occupy the same volume, so the molar amounts may be replaced with molar concentrations

The below graph illustrates the change in concentrations over time for the decomposition of ammonia into nitrogen and hydrogen at 1100 °C. Slopes of the tangent lines at t = 500 s show that the instantaneous rates derived from all three species involved in the reaction are related by their stoichiometric factors. The rate of hydrogen production, for example, is observed to be three times greater than that for nitrogen production:

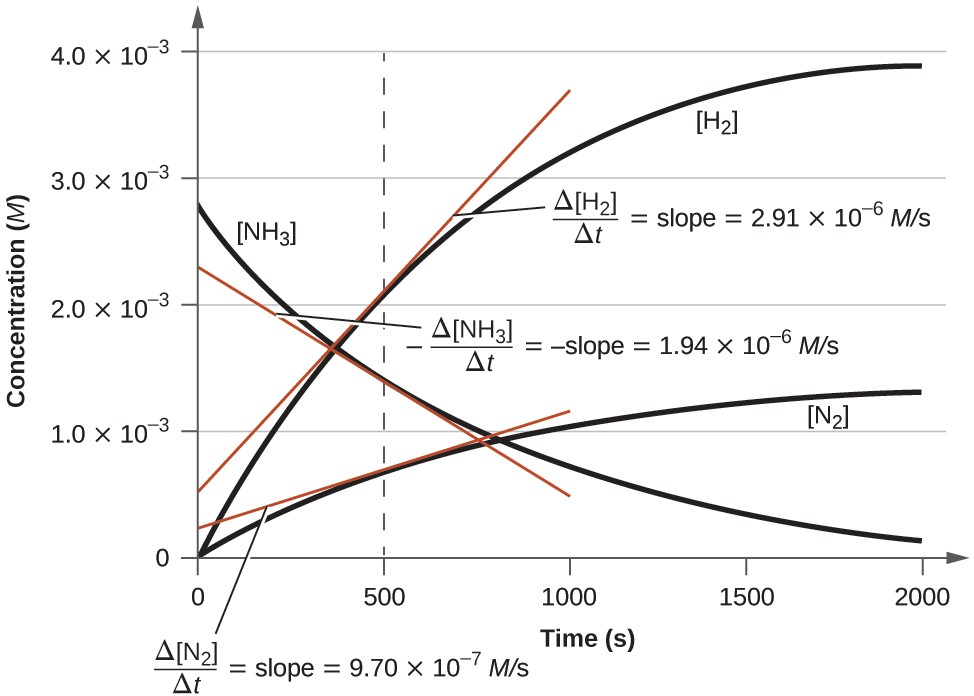
Changes in concentrations of the reactant and products for the reaction 2NH3 (g) —–à N2 (g) + 3 H2 (g)
The rates of change of the three concentrations are related by the reaction stoichiometry, as shown by the different slopes of the tangents at t = 500 s
Factors Affecting Reaction Rates
The rates at which reactants are consumed and products are formed during chemical reactions vary greatly. Five factors typically affecting the rates of chemical reactions will be explored in this section: the chemical nature of the reacting substances, the state of subdivision (one large lump versus many small particles) of the reactants, the temperature of the reactants, the concentration of the reactants, and the presence of a catalyst
The Chemical Nature of the Reacting Substances
The rate of a reaction depends on the nature of the participating substances. Reactions that appear similar may have different rates under the same conditions, depending on the identity of the reactants. For example, when small pieces of the metals iron and sodium are exposed to air, the sodium reacts completely with air overnight, whereas the iron is barely affected. The active metals calcium and sodium both react with water to form hydrogen gas and a base. Yet calcium reacts at a moderate rate, whereas sodium reacts so rapidly that the reaction is almost explosive
The Physical States of the Reactants
A chemical reaction between two or more substances requires intimate contact between the reactants. When reactants are in different physical states, or phases (solid, liquid, gaseous, dissolved), the reaction takes place only at the interface between the phases. Consider the heterogeneous reaction between a solid phase and either a liquid or gaseous phase. Compared with the reaction rate for large solid particles, the rate for smaller particles will be greater because the surface area in contact with the other reactant phase is greater. For example, large pieces of iron react more slowly with acids than they do with finely divided iron powder (Figure below ). Large pieces of wood smolder, smaller pieces burn rapidly, and saw dust burns explosively

(a) Iron powder reacts rapidly with dilute hydrochloric acid and produces bubbles of hydrogen gas: 2Fe(s) + 6HCl(aq) ———à 2FeCl3(aq) + 3H2(g). (b) An iron nail reacts more slowly because the surface area exposed to the acid is much less.
Temperature of the Reactants
Chemical reactions typically occur faster at higher temperatures. Food can spoil quickly when left on the kitchen counter. However, the lower temperature inside of a refrigerator slows that process so that the same food remains fresh for days. Gas burners, hot plates, and ovens are often used in the laboratory to increase the speed of reactions that proceed slowly at ordinary temperatures. For many chemical processes, reaction rates are approximately doubled when the temperature is raised by 10 °C.
Concentrations of the Reactants
The rates of many reactions depend on the concentrations of the reactants. Rates usually increase when the concentration of one or more of the reactants increases. For example, calcium carbonate (CaCO3) deteriorates as a result of its reaction with the pollutant sulfur dioxide. The rate of this reaction depends on the amount of sulfur dioxide in the air (Figure 17.7). An acidic oxide, sulfur dioxide combines with water vapor in the air to produce sulfurous acid in the following reaction :

In a polluted atmosphere where the concentration of sulfur dioxide is high, calcium carbonate deteriorates more rapidly than in less polluted air. Similarly, phosphorus burns much more rapidly in an atmosphere of pure oxygen than in air, which is only about 20% oxygen

Statues made from carbonate compounds such as limestone and marble typically weather slowly over time due to the actions of water, and thermal expansion and contraction. However, pollutants like sulfur dioxide can accelerate weathering. As the concentration of air pollutants increases, deterioration of limestone occurs more rapidly. (credit: James P Fisher III)
The Presence of a Catalyst
Relatively dilute aqueous solutions of hydrogen peroxide, H2O2, are commonly used as topical antiseptics. Hydrogen peroxide decomposes to yield water and oxygen gas according to the equation :
2 H2 O2 —-à 2 H2O (l) + O2 (g)
Under typical conditions, this decomposition occurs very slowly. When dilute H2O2(aq) is poured onto an open wound, however, the reaction occurs rapidly and the solution foams because of the vigorous production of oxygen gas. This dramatic difference is caused by the presence of substances within the wound’s exposed tissues that accelerate the decomposition process. Substances that function to increase the rate of a reaction are called catalysts .
Rate Laws
The rate of a reaction is often affected by the concentrations of reactants. Rate laws (sometimes called differential rate laws) or rate equations are mathematical expressions that describe the relationship between the rate of a chemical reaction and the concentration of its reactants. As an example, consider the reaction described by the chemical equation
aA + bB ———- Products
where a and b are stoichiometric coefficients. The rate law for this reaction is written as:
rate = k [ A ] m [ B ]
in which [A] and [B] represent the molar concentrations of reactants, and k is the rate constant, which is specific for a particular reaction at a particular temperature. The exponents m and n are the reaction orders and are typically positive integers, though they can be fractions, negative, or zero. The rate constant k and the reaction orders m and n must be determined experimentally by observing how the rate of a reaction changes as the concentrations of the reactants are changed. The rate constant k is independent of the reactant concentrations, but it does vary with temperature.
The reaction orders in a rate law describe the mathematical dependence of the rate on reactant concentrations. Referring to the generic rate law above, the reaction is m order with respect to A and n order with respect to B. For example, if m = 1 and n = 2, the reaction is first order in A and second order in B. The overall reaction order is simply the sum of orders for each reactant. For the example rate law here, the reaction is third order overall (1 + 2 = 3). A few specific examples are shown below to further illustrate this concept.
The rate law:
rate=k[H2O2]
describes a reaction that is first order in hydrogen peroxide and first order overall. The rate law:
rate=k[C4H6]2
describes a reaction that is second order in C4H6 and second order overall. The rate law:
rate=k[H+][OH−]
describes a reaction that is first order in H+, first order in OH−, and second order overall.
Reaction Order and Rate Constant Units
In some cases , the reaction orders in the rate law happen to be the same as the coefficients in the chemical equation for the reaction. This is merely a coincidence and very often not the case
Rate laws may exhibit fractional orders for some reactants, and negative reaction orders are sometimes observed when an increase in the concentration of one reactant causes a decrease in reaction rate. A few examples illustrating these points are provided

It is important to note that rate laws are determined by experiment only and are not reliably predicted by reaction stoichiometry

Note that the units in this table were derived using specific units for concentration (mol/L) and time (s), though any valid units for these two properties may be used.
Integrated Rate Laws
The rate laws discussed thus far relate the rate and the concentrations of reactants. We can also determine a second form of each rate law that relates the concentrations of reactants and time. These are called integrated rate laws. We can use an integrated rate law to determine the amount of reactant or product present after a period of time or to estimate the time required for a reaction to proceed to a certain extent. For example, an integrated rate law is used to determine the length of time a radioactive material must be stored for its radioactivity to decay to a safe level.
Using calculus, the differential rate law for a chemical reaction can be integrated with respect to time to give an equation that relates the amount of reactant or product present in a reaction mixture to the elapsed time of the reaction. This process can either be very straightforward or very complex, depending on the complexity of the differential rate law. For purposes of discussion, we will focus on the resulting integrated rate laws for first-
, second-, and zero-order reactions
First-Order Reactions
Integration of the rate law for a simple first-order reaction (rate = k[A]) results in an equation describing how the reactant concentration varies with time
[A]t = [A]0 e-kt
where [A]t is the concentration of A at any time t, [A]0 is the initial concentration of A, and k is the first-order rate constant
For mathematical convenience, this equation may be rearranged to other formats, including direct and indirect proportionalities :
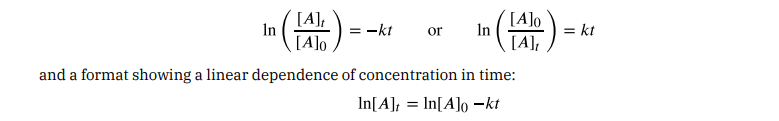
Second-Order Reactions
The equations that relate the concentrations of reactants and the rate constant of second-order reactions can be fairly complicated. To illustrate the point with minimal complexity, only the simplest second-order reactions will be described here, namely, those whose rates depend on the concentration of just one reactant. For these types of reactions, the differential rate law is written as:
rate = k [A]2

The integrated rate law for second-order reactions has the form of the equation of a straight line:

Zero-Order Reactions
For zero-order reactions, the differential rate law is:
rate = k
A zero-order reaction thus exhibits a constant reaction rate, regardless of the concentration of its reactant(s). This may seem counterintuitive, since the reaction rate certainly can’t be finite when the reactant concentration is zero. For purposes of this introductory text, it will suffice to note that zero-order kinetics are observed for some reactions only under certain specific conditions. These same reactions exhibit different kinetic behaviors when the specific conditions aren’t met, and for this reason the more prudent term pseudozero-order is sometimes used. The integrated rate law for a zero-order reaction is a linear function:
[A]t = -kt = [A]0
y = mx + b
A plot of [A] versus t for a zero-order reaction is a straight line with a slope of −k and a y-intercept of [A]0. Below graph shows a plot of [NH3] versus t for the thermal decomposition of ammonia at the surface of two different heated solids. The decomposition reaction exhibits first-order behavior at a quartz (SiO2) surface, as suggested by the exponentially decaying plot of concentration versus time. On a tungsten surface, however, the plot is linear, indicating zero-order kinetics.
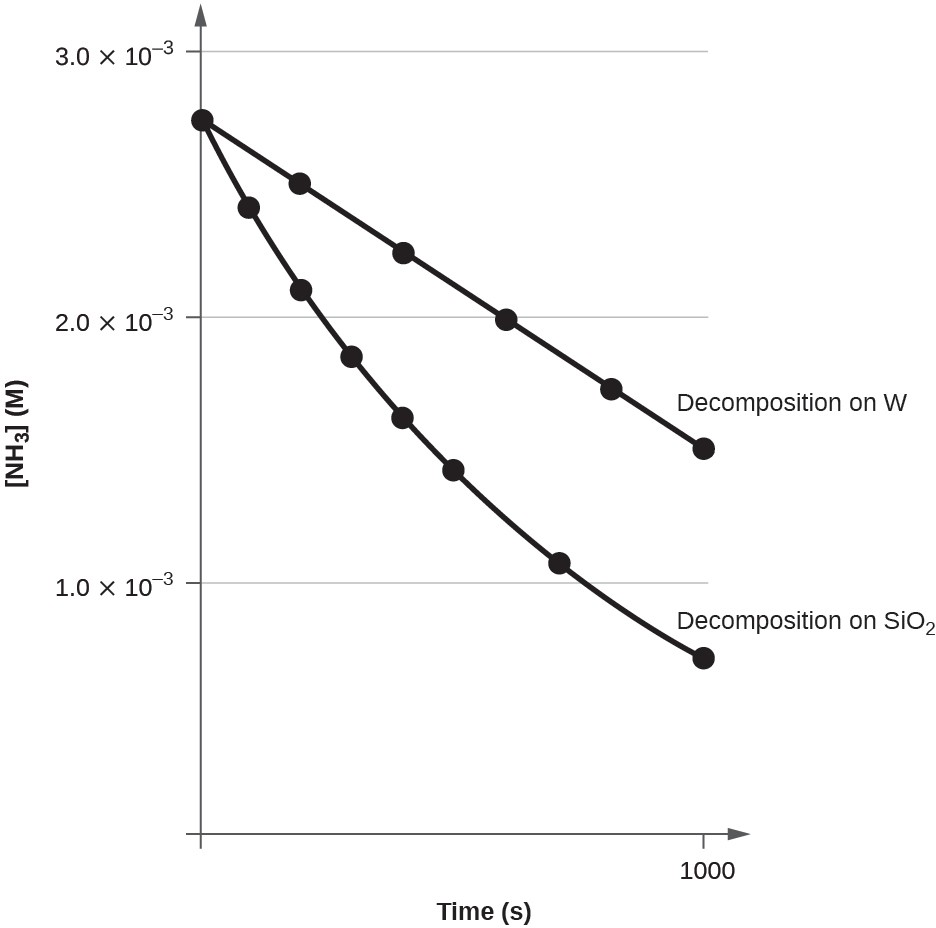
The decomposition of NH3 on a tungsten (W) surface is a zero-order reaction, whereas on a quartz (SiO2) surface, the reaction is first order
